FDA rejections threaten lives of children with rare diseases
FDA rejections threaten lives of children with rare diseases
- Parents of children with rare diseases expressed fears regarding the FDA's rejections of necessary treatments.
- Over 100 mothers staged a funeral outside the FDA, demanding access to therapies for life-threatening conditions.
- The FDA's refusal to approve drugs has led to calls for accountability and urgent action from Congress.
Story
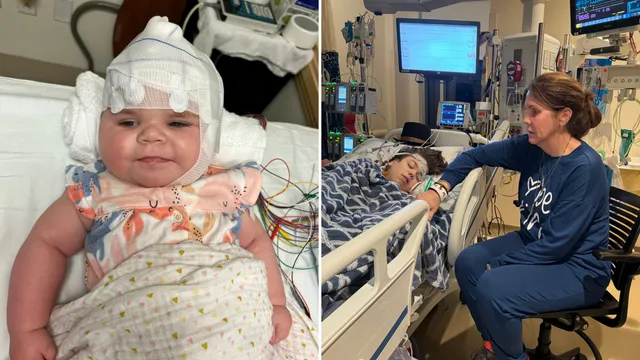
In the United States, a growing dissatisfaction has emerged within the rare disease community following the FDA's rejection of several crucial treatment drugs. Numerous mothers and advocates organized a demonstration outside the FDA’s headquarters to protest against the agency’s denial of treatments for severe conditions, particularly those affecting children. The event, symbolically staged as a funeral, highlighted the fears of parents who feel their children's lives are at stake due to lack of approved therapies. This protest garnered significant media attention and raised awareness of the urgent need for action. Kim Higbee, board member of the Hope For PDCD Foundation, revealed that as of early January 2025, the FDA had issued 23 complete response letters, indicating no approvals for currently tested rare disease drugs, often resulting in rejections or delays pending further studies. This situation particularly impacts children suffering from diseases such as pyruvate dehydrogenase deficiency (PDCD) and Hunter syndrome, conditions that without proper treatments can lead to life-threatening complications. Higbee's daughter, Harlow, is part of this community, having benefited from a clinical trial for a potential treatment. However, the recent denial of the ongoing drug trial has instilled a sense of despair among families who have watched their children improve on these therapies. Parents expressed their trauma and frustrations through poignant testimonials, sharing fears of potential losses, as highlighted by the experiences of mothers like Kim Stephens, whose son lives with Hunter syndrome. The FDA's stringent requirements and its apparent repudiation of alternative approval pathways come amidst increasing calls for the agency to reconsider its stance on expedited treatments. Advocacy leaders, including Senator Ron Johnson, are calling for investigations and accountability from the FDA, demanding that the approval process for these life-altering therapies become more lenient, allowing children access to potentially life-saving innovations.