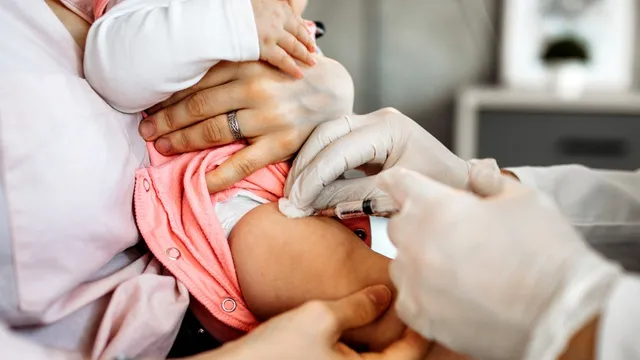
CDC advisers debate reversal of newborn hepatitis B vaccine recommendation
CDC advisers debate reversal of newborn hepatitis B vaccine recommendation
- The CDC's ACIP is planning a vote on recommendations for the hepatitis B vaccine for newborns.
- Research indicates that the vaccine has successfully prevented millions of infections and hospitalizations.
- The outcome of the vote could influence healthcare practices and insurance coverage for newborn vaccinations.
Story
In the United States, a group of advisers to the Centers for Disease Control and Prevention (CDC) is set to vote on whether to discontinue the long-standing recommendation that every newborn receive the hepatitis B vaccine within 24 hours of birth. This vaccine, which protects against a virus that can lead to serious liver disease and high mortality rates, has faced skepticism from some groups questioning its safety and efficacy. Analysis reveals that despite the successful prevention of millions of infections and hospitalizations, debates surrounding vaccination practices persist, prompting the ACIP's upcoming meeting. The vaccine’s birth dose is particularly important as hepatitis B can be transmitted from mother to child during childbirth, putting infants at risk. The advisory committee has recently seen changes in leadership, with Milhoan appointed as the new chair, suggesting a shift in perspectives on vaccination. While previous chair Martin Kuldorff expressed doubts about childhood vaccines, including hepatitis B, the new leadership aims to consider the clinical assessment of individual infants regarding their mothers' risk factors, rather than maintaining a blanket recommendation. Experts have also weighed in, with pediatric infectious disease expert Dr. Sean O’Leary noting a lack of serious adverse reactions tied to the vaccine after numerous administrations to newborns. This is further supported by a comprehensive review of over 400 studies, which concluded that no significant health issues stem from the vaccine's administration, reinforcing its importance in public health. The upcoming vote has attracted considerable attention as it may influence insurance coverage for the vaccine, thus affecting access for infants at risk of infection. This decision comes after earlier discussions were postponed due to confusion among committee members. As the meeting approaches, conflicting opinions on the best practices for vaccination remain a focal point, highlighting a continual need for clear communication and comprehensive care in prenatal and infant health.